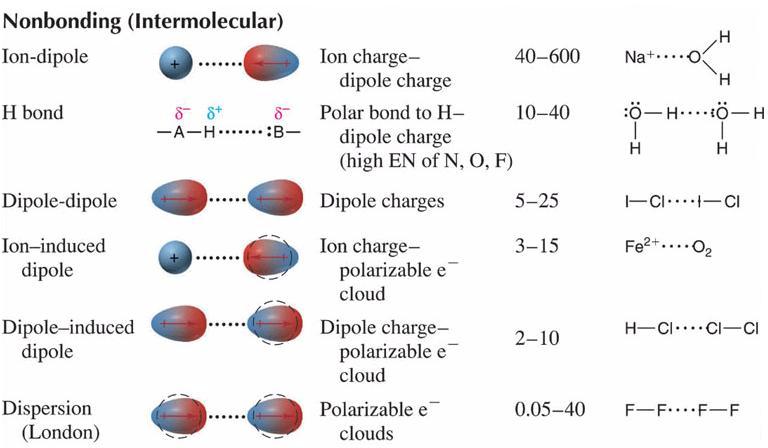
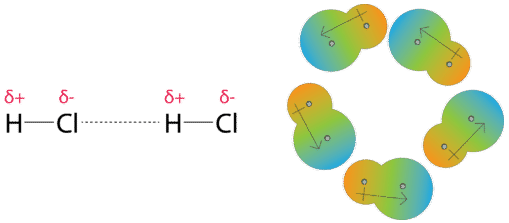1 Chapter 10 Liquids and Solids. 2 3 Van Der Waals Forces These are intermolecular forces of attraction between neutral molecules. The Nobel Prize in. - ppt download
are the four types of van der waals forces as follows 1. london dispersion forces 2.dipole dipole forces 3.dipole induced dipole 4.hydrogen bonding ?

Vanderwaals Interactions | London forces | structure and bonding | bsc 1st year chemistry |chemboost - YouTube

TOPIC 4: CHEMICAL BONDING & STRUCTURE. ESSENTIAL IDEA The physical properties of molecular substances result from different types of forces between their. - ppt download