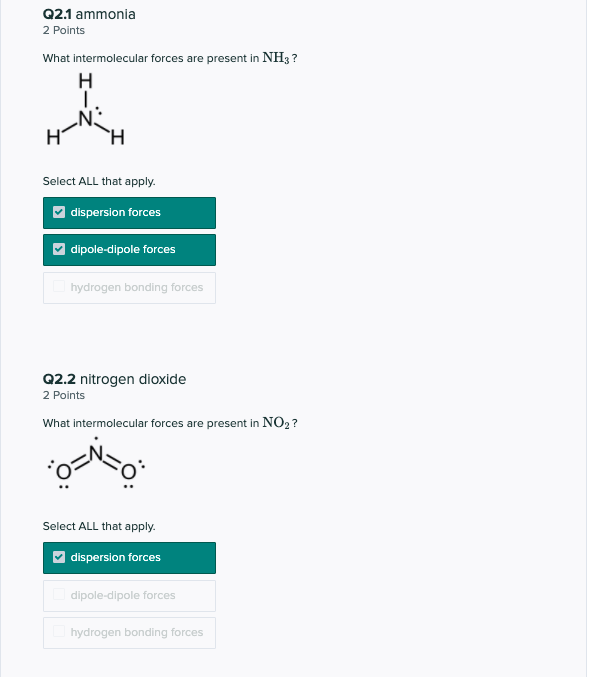
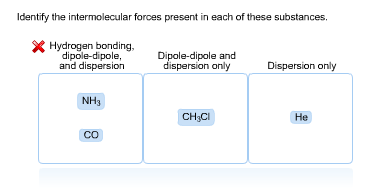
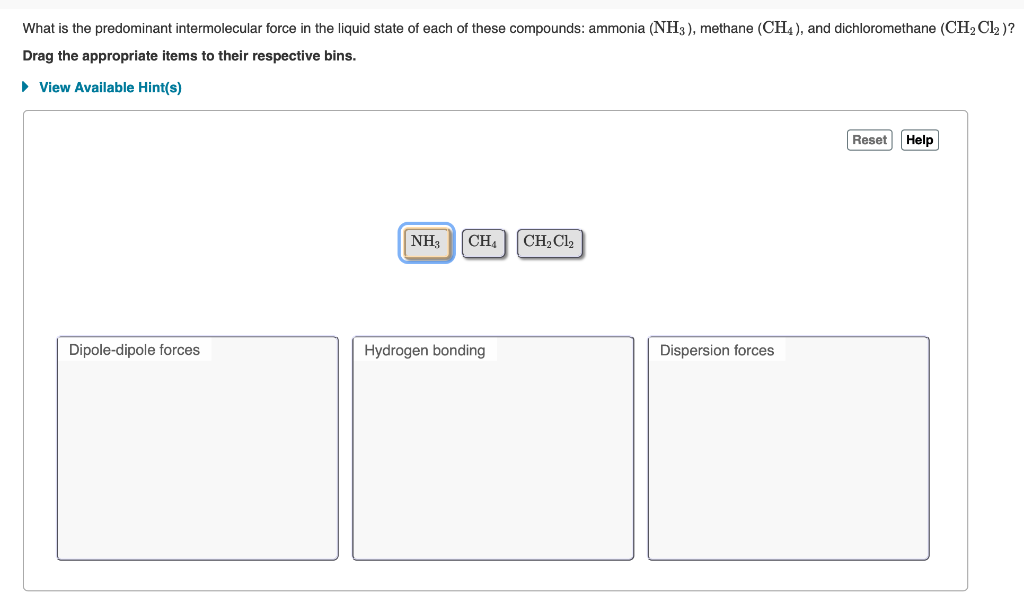
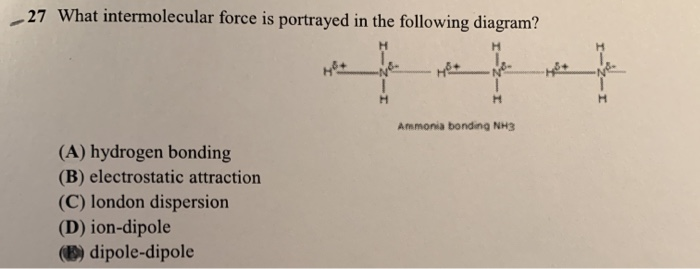
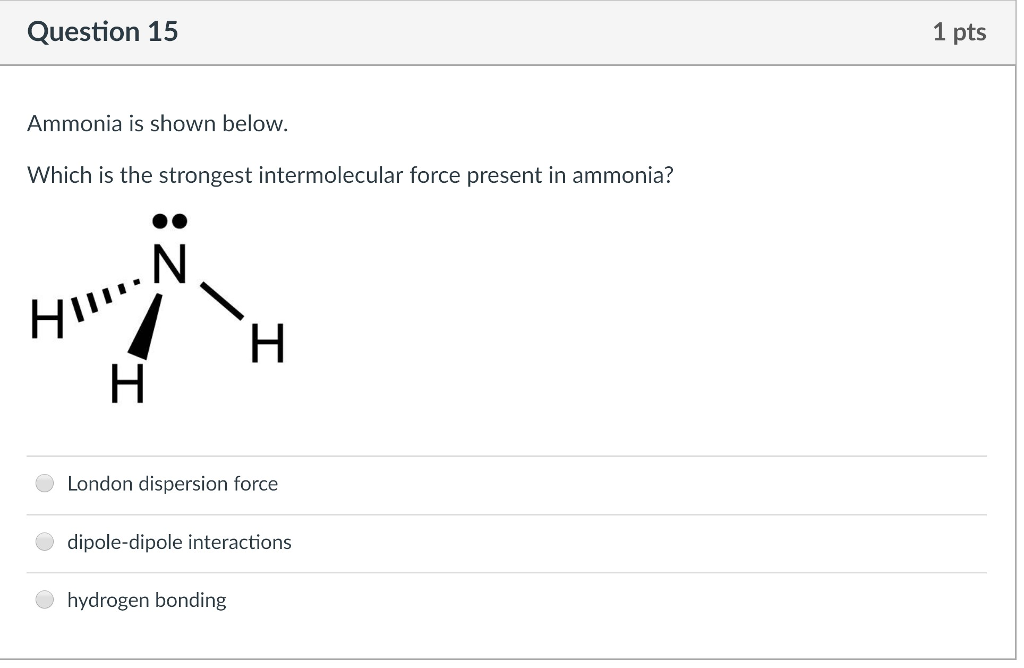
Compare and contrast the compounds NH3 and PH3 in terms of bonding, molecular geometry (shape), and intermolecular forces. Which substance would experience a stronger attraction between its molecules? Provide a straight forward
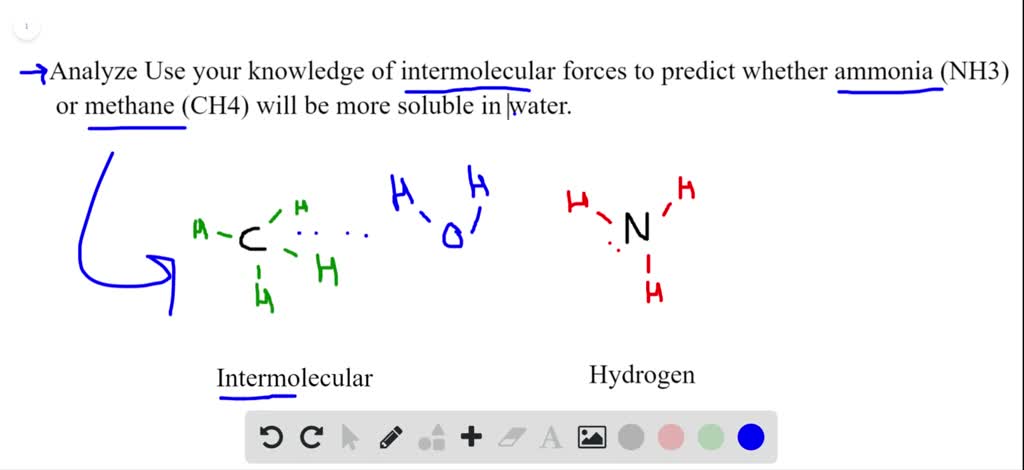
SOLVED:Analyze Use your knowledge of intermolecular forces to predict whether ammonia (NH 3 ) or methane (CH4) will be more soluble in water.

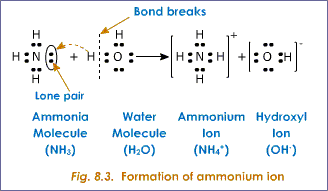
SOLVED: What types of intermolecular forces exist between NH3 and H2O? Select all that apply. dipole induced dipole dipole dipole hydrogen bond London dispersion